Chain of Custody Protocols for Blood Culture Samples in Medical Diagnostic Labs
Summary
- Proper labeling of Blood Culture samples is essential in maintaining the chain of custody.
- Strict documentation and tracking processes should be followed to prevent any errors or mishandling of samples.
- Routine training and education for lab staff on chain of custody protocols is crucial in ensuring compliance and accuracy.
Introduction
When dealing with Blood Culture samples in a medical diagnostic lab, it is crucial to follow specific protocols to ensure the proper chain of custody. Chain of custody refers to the chronological documentation or paper trail that shows the seizure, custody, control, transfer, and analysis of a sample, ensuring that its integrity is maintained throughout the process. In the United States, healthcare facilities, including clinical labs and hospitals, must adhere to strict protocols to prevent contamination, mix-ups, or mishandling of Blood Culture samples. In this article, we will explore the protocols that should be followed to maintain the chain of custody for Blood Culture samples in a medical diagnostic lab.
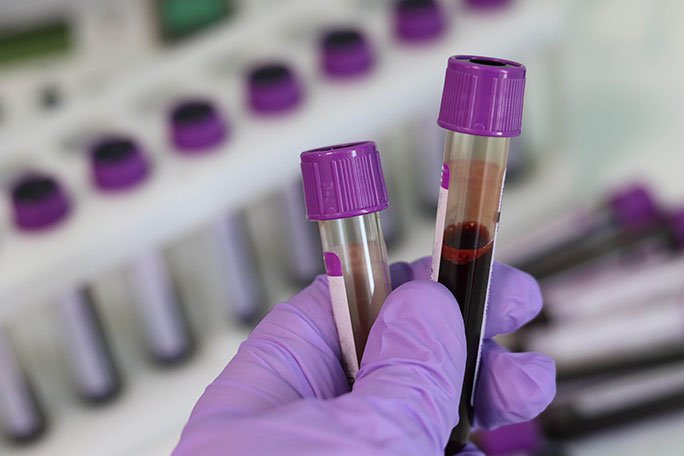
Proper Labeling of Samples
One of the most critical aspects of maintaining the chain of custody for Blood Culture samples is proper labeling. Each sample should be clearly labeled with the patient's name, date of collection, unique identifier (such as a barcode), and the initials of the person collecting the sample. Additionally, the label should include any relevant clinical information, such as the reason for the test, the type of specimen, and any special instructions for handling.
Key Points:
- Ensure that labels are securely attached to the sample container and that they are legible and not damaged.
- Use waterproof and durable labels to prevent fading or smudging during transport and storage.
- Double-check the information on the label against the requisition form to ensure accuracy.
Strict Documentation and Tracking
In addition to proper labeling, strict documentation and tracking processes should be followed to maintain the chain of custody for Blood Culture samples. This includes documenting the date and time of sample collection, the identity of the person collecting the sample, and any relevant observations or special circumstances. A log should be kept to track the movement of samples from collection to analysis, including transfers between different departments or facilities.
Key Points:
- Use a secure and centralized system to document all sample-related information, including electronic databases or paper logs.
- Implement protocols for sample storage, transport, and disposal to prevent contamination or tampering.
- Perform regular audits and reviews of documentation to identify any Discrepancies or errors in the chain of custody.
Training and Education
Ensuring compliance with chain of custody protocols requires routine training and education for lab staff involved in handling Blood Culture samples. All personnel should be familiar with the proper procedures for sample collection, labeling, documentation, and tracking to minimize the risk of errors or mishandling. Training should be provided to new employees and as part of ongoing professional development programs for existing staff.
Key Points:
- Provide comprehensive training on chain of custody protocols, including the importance of maintaining Sample Integrity and the consequences of non-compliance.
- Offer refresher courses and updates on any changes to protocols or Regulations related to Sample Handling.
- Encourage open communication and reporting of any issues or concerns related to the chain of custody to prevent errors or Quality Control issues.
Conclusion
Proper chain of custody protocols are essential for ensuring the accuracy and reliability of Blood Culture samples in a medical diagnostic lab. By following strict procedures for labeling, documentation, tracking, and staff training, healthcare facilities can maintain the integrity of samples and prevent errors or mishandling. Compliance with these protocols not only ensures the quality of diagnostic testing but also upholds patient safety and trust in the healthcare system.
Disclaimer: The content provided on this blog is for informational purposes only, reflecting the personal opinions and insights of the author(s) on phlebotomy practices and healthcare. The information provided should not be used for diagnosing or treating a health problem or disease, and those seeking personal medical advice should consult with a licensed physician. Always seek the advice of your doctor or other qualified health provider regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website. If you think you may have a medical emergency, call 911 or go to the nearest emergency room immediately. No physician-patient relationship is created by this web site or its use. No contributors to this web site make any representations, express or implied, with respect to the information provided herein or to its use. While we strive to share accurate and up-to-date information, we cannot guarantee the completeness, reliability, or accuracy of the content. The blog may also include links to external websites and resources for the convenience of our readers. Please note that linking to other sites does not imply endorsement of their content, practices, or services by us. Readers should use their discretion and judgment while exploring any external links and resources mentioned on this blog.
