Ensuring Proper Sterilization of Synovial Fluid Sample Collection Equipment in Medical Diagnostic Labs
Summary
- Sterilization of synovial fluid sample collection equipment is crucial in preventing infections and ensuring accurate Test Results.
- The frequency of sterilization of equipment in a medical diagnostic lab in the United States should adhere to strict guidelines set by regulatory bodies.
- Regular maintenance and proper sterilization can help healthcare facilities maintain high standards of quality and safety in sample collection processes.
Introduction
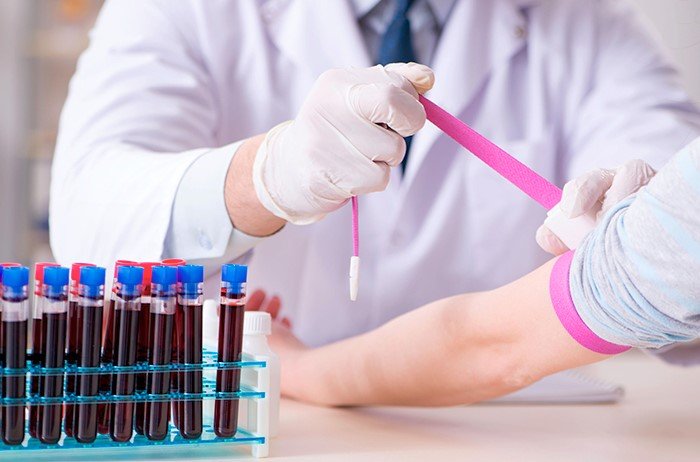
Medical Diagnostic Labs play a critical role in the healthcare system by providing accurate and timely Test Results that aid in diagnosing and monitoring various medical conditions. Synovial fluid sample collection is a common procedure performed in these labs to aid in the diagnosis of joint-related issues such as arthritis and infections. It is essential to ensure that the equipment used in collecting synovial fluid samples is properly sterilized to prevent contamination and ensure accurate Test Results.
Why Sterilization is Important
Sterilization of equipment, including needles, syringes, and collection tubes, is crucial in preventing infections in patients undergoing synovial fluid sample collection. Contaminated equipment can introduce bacteria or other pathogens into the sample, leading to false-positive or false-negative Test Results. This can have serious implications for patient care, as an inaccurate diagnosis can result in inappropriate treatment or delayed intervention.
Risks of Improper Sterilization
- Risk of introducing infections: Improperly sterilized equipment can introduce bacteria, viruses, or fungi into the synovial fluid sample, leading to infections in the patient.
- Inaccurate Test Results: Contaminated equipment can compromise the integrity of the sample, leading to inaccurate Test Results and potentially incorrect diagnosis and treatment.
- Potential harm to patients: Infections or inaccurate diagnoses resulting from improper sterilization can harm patients and compromise their health outcomes.
Regulatory Guidelines
In the United States, healthcare facilities, including medical Diagnostic Labs, are required to adhere to strict guidelines set by regulatory bodies such as the Centers for Disease Control and Prevention (CDC) and the Occupational Safety and Health Administration (OSHA). These guidelines outline the proper procedures for the sterilization of medical equipment to ensure patient safety and prevent infections.
FDA Regulations
The Food and Drug Administration (FDA) also sets specific Regulations for medical devices, including equipment used in sample collection procedures. Healthcare facilities must comply with FDA Regulations to ensure the safety and effectiveness of the equipment used in synovial fluid sample collection.
Recommended Practices
- Regular maintenance: Healthcare facilities should regularly inspect and maintain equipment used in synovial fluid sample collection to ensure proper functioning and prevent contamination.
- Proper sterilization techniques: Equipment should be sterilized using appropriate techniques such as autoclaving, chemical disinfection, or ethylene oxide sterilization.
- Documentation: Healthcare facilities should maintain documentation of equipment sterilization processes to ensure compliance with regulatory guidelines.
Frequency of Sterilization
The frequency of sterilization of synovial fluid sample collection equipment in a medical diagnostic lab in the United States depends on several factors, including the type of equipment used, the volume of samples processed, and the risk of contamination. While there is no one-size-fits-all answer, healthcare facilities should follow general guidelines to ensure the safety and effectiveness of sample collection procedures.
General Recommendations
- Single-use equipment: Disposable needles and syringes should be used for each patient to prevent the risk of contamination. These items should be disposed of properly after each use.
- Reusable equipment: Equipment that can be sterilized and reused, such as collection tubes and trays, should be thoroughly cleaned and sterilized after each use.
- High-risk procedures: In cases where there is a higher risk of contamination, such as samples obtained from immunocompromised patients, equipment should be sterilized more frequently.
Best Practices
- Establish a sterilization schedule: Healthcare facilities should develop a sterilization schedule based on the volume of samples processed and the risk of contamination. This schedule should be regularly reviewed and updated as needed.
- Quality Control measures: Regularly monitor and assess the effectiveness of the sterilization process to ensure that equipment is properly sterilized and free from contamination.
- Training and education: Provide staff members with training on proper sterilization techniques and infection control practices to help prevent contamination and ensure patient safety.
Conclusion
Ensuring the proper sterilization of synovial fluid sample collection equipment in a medical diagnostic lab is essential for maintaining high standards of quality and safety in healthcare. Healthcare facilities should adhere to regulatory guidelines and best practices to prevent infections, ensure accurate Test Results, and protect patient health. By establishing a sterilization schedule, implementing Quality Control measures, and providing staff education, healthcare facilities can promote a culture of safety and excellence in sample collection processes.
Disclaimer: The content provided on this blog is for informational purposes only, reflecting the personal opinions and insights of the author(s) on phlebotomy practices and healthcare. The information provided should not be used for diagnosing or treating a health problem or disease, and those seeking personal medical advice should consult with a licensed physician. Always seek the advice of your doctor or other qualified health provider regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website. If you think you may have a medical emergency, call 911 or go to the nearest emergency room immediately. No physician-patient relationship is created by this web site or its use. No contributors to this web site make any representations, express or implied, with respect to the information provided herein or to its use. While we strive to share accurate and up-to-date information, we cannot guarantee the completeness, reliability, or accuracy of the content. The blog may also include links to external websites and resources for the convenience of our readers. Please note that linking to other sites does not imply endorsement of their content, practices, or services by us. Readers should use their discretion and judgment while exploring any external links and resources mentioned on this blog.
