Best Practices for Quality Control in Phlebotomy Procedures in the United States
Summary
- Establishing Quality Control measures is essential in ensuring accurate and reliable phlebotomy procedures in the United States.
- Maintaining proper training and certification for phlebotomists is crucial for upholding Quality Control standards in clinical labs and hospitals.
- Regular audits, feedback mechanisms, and continuous improvement initiatives are key to sustaining Quality Control in phlebotomy procedures.
Introduction
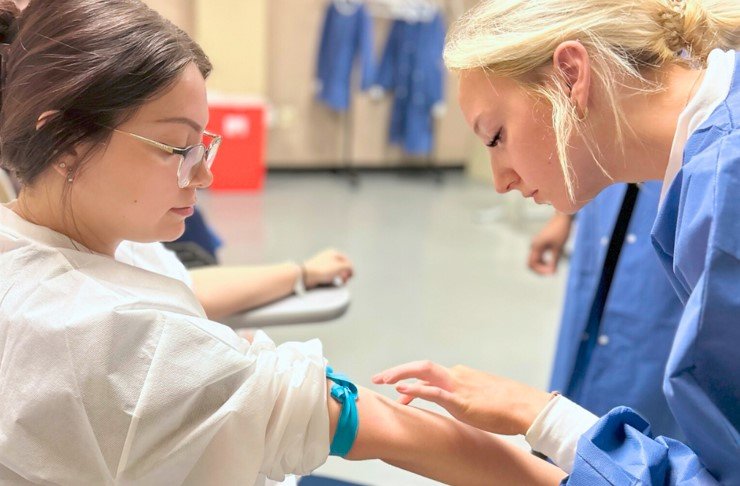
Phlebotomy procedures are an integral part of healthcare services in the United States, involving the collection of blood samples for diagnostic testing and monitoring patients' health. To ensure the accuracy, reliability, and safety of these procedures, it is crucial to establish and maintain Quality Control measures in clinical labs and hospitals. In this article, we will explore the best practices for upholding Quality Control standards in phlebotomy procedures.
Training and Certification
Proper training and certification of phlebotomists are essential for maintaining Quality Control in phlebotomy procedures. Certified phlebotomists undergo rigorous training programs that cover anatomy, physiology, specimen collection techniques, safety protocols, and Regulations. By ensuring that phlebotomists are well-trained and certified, healthcare facilities can uphold Quality Control standards and minimize errors in blood sample collection.
Best Practices:
- Ensure that all phlebotomists are certified by accredited organizations such as the American Society for Clinical Pathology (ASCP) or the National Healthcareer Association (NHA).
- Provide ongoing training and professional development opportunities to keep phlebotomists updated on the latest techniques and best practices in phlebotomy.
- Implement regular competency assessments to evaluate phlebotomists' skills and knowledge and address any areas for improvement.
Standard Operating Procedures
Developing and enforcing standard operating procedures (SOPs) for phlebotomy procedures is crucial for ensuring consistency, efficiency, and Quality Control in clinical labs and hospitals. SOPs outline the step-by-step guidelines for collecting blood samples, handling specimens, and maintaining a sterile and safe environment during phlebotomy procedures. By following SOPs, phlebotomists can minimize errors, reduce contamination risks, and enhance the overall quality of blood sample collection.
Best Practices:
- Create clear and detailed SOPs that cover all aspects of phlebotomy procedures, including patient identification, specimen labeling, vein selection, and sample transport.
- Train phlebotomists on SOPs and ensure that they understand and comply with the established guidelines for blood collection and handling.
- Regularly review and update SOPs to reflect the latest industry standards, technology advancements, and regulatory requirements.
Quality Control Audits
Conducting regular Quality Control audits is essential for monitoring the performance and compliance of phlebotomy procedures in clinical labs and hospitals. Quality Control audits involve assessing the accuracy, precision, and reliability of blood sample collection processes, identifying any deviations or errors, and implementing corrective actions to improve performance. By proactively conducting audits, healthcare facilities can detect issues early, prevent errors, and maintain high standards of Quality Control in phlebotomy procedures.
Best Practices:
- Establish a Quality Control team responsible for conducting routine audits, analyzing data, and reporting findings to management.
- Use Quality Control software and tools to automate audit processes, track performance metrics, and generate reports for continuous improvement.
- Implement feedback mechanisms to gather input from phlebotomists, Healthcare Providers, and patients on the quality of phlebotomy procedures and areas for enhancement.
Continuous Improvement Initiatives
Implementing continuous improvement initiatives is key to sustaining Quality Control measures in phlebotomy procedures. Continuous improvement involves identifying opportunities for enhancing processes, addressing performance gaps, and implementing changes to achieve better outcomes. By fostering a culture of continuous improvement, healthcare facilities can proactively respond to challenges, adapt to evolving standards, and deliver high-quality phlebotomy services to patients.
Best Practices:
- Encourage phlebotomists to share feedback, suggestions, and innovative ideas for improving phlebotomy procedures and Workflow efficiency.
- Collaborate with cross-functional teams, including nurses, laboratory technicians, and quality assurance specialists, to identify process improvements and implement best practices.
- Monitor key performance indicators, such as turnaround times, specimen quality, and Patient Satisfaction, to measure the impact of continuous improvement initiatives on Quality Control in phlebotomy procedures.
Conclusion
Establishing and maintaining Quality Control measures in phlebotomy procedures is essential for ensuring the accuracy, reliability, and safety of blood sample collection in clinical labs and hospitals. By implementing best practices such as proper training and certification, standard operating procedures, Quality Control audits, and continuous improvement initiatives, healthcare facilities can uphold high standards of Quality Control and deliver superior phlebotomy services to patients.
Disclaimer: The content provided on this blog is for informational purposes only, reflecting the personal opinions and insights of the author(s) on the topics. The information provided should not be used for diagnosing or treating a health problem or disease, and those seeking personal medical advice should consult with a licensed physician. Always seek the advice of your doctor or other qualified health provider regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website. If you think you may have a medical emergency, call 911 or go to the nearest emergency room immediately. No physician-patient relationship is created by this web site or its use. No contributors to this web site make any representations, express or implied, with respect to the information provided herein or to its use. While we strive to share accurate and up-to-date information, we cannot guarantee the completeness, reliability, or accuracy of the content. The blog may also include links to external websites and resources for the convenience of our readers. Please note that linking to other sites does not imply endorsement of their content, practices, or services by us. Readers should use their discretion and judgment while exploring any external links and resources mentioned on this blog.
