Maintaining the Chain of Custody during Phlebotomy Procedures: Essential Steps and Best Practices
Summary
- Properly identify the patient before starting the phlebotomy procedure.
- Label the blood samples accurately and immediately after collection.
- Ensure proper storage and transportation of the blood samples to maintain the chain of custody.
Introduction
Phlebotomy is a critical process in healthcare that involves drawing blood samples from patients for diagnostic testing. Maintaining the chain of custody during phlebotomy procedures is crucial to ensure the integrity of the samples and accurate Test Results. In the United States, specific steps are followed to preserve the chain of custody during phlebotomy. In this article, we will discuss the essential steps involved in maintaining the chain of custody during phlebotomy procedures in the context of home healthcare, clinical labs, and hospitals.
Proper Patient Identification
Before starting the phlebotomy procedure, it is essential to properly identify the patient to ensure that the blood samples are collected from the right individual. This step involves verifying the patient's full name, date of birth, and other identifying information such as a unique identifier like a medical record number. In home healthcare settings, phlebotomists must confirm the patient's identity by checking their identification card or asking for other proof of identity.
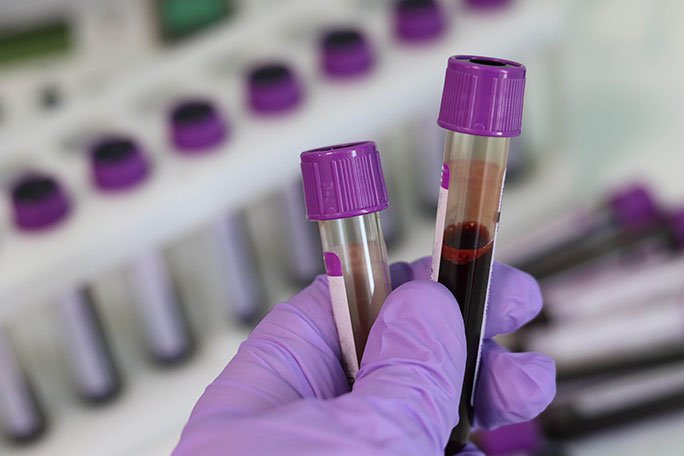
Labeling of Blood Samples
After collecting the blood samples, the next crucial step in maintaining the chain of custody is labeling the samples accurately and immediately. Each blood sample must be labeled with the patient's full name, date of birth, unique identifier, date and time of collection, and the phlebotomist's initials. In clinical labs and hospitals, barcoding technology is often used to label blood samples to minimize the risk of errors in labeling. Proper labeling ensures that the samples can be traced back to the correct patient and prevents mix-ups during processing.
Secure Storage and Transportation
Once the blood samples are collected and labeled, they must be stored and transported securely to maintain the chain of custody. In clinical labs and hospitals, blood samples are typically stored in designated refrigerators or freezers at the appropriate temperature to preserve their integrity. During transportation, the samples must be handled carefully to prevent damage or contamination. Phlebotomists must follow protocols for transporting blood samples, such as using sealed containers or biohazard bags to ensure the samples reach the testing facility safely.
Chain of Custody Documentation
Documentation is a critical aspect of maintaining the chain of custody during phlebotomy procedures. Phlebotomists are required to record every step of the process, from patient identification to sample collection, labeling, storage, and transportation. Detailed documentation helps track the movement of the blood samples and provides a clear record of who handled the samples at each stage. In the event of any Discrepancies or issues with the samples, having thorough documentation can help identify potential errors and ensure the integrity of the testing process.
Quality Control Measures
To maintain the chain of custody during phlebotomy procedures, Quality Control measures are implemented in clinical labs and hospitals. These measures include regular audits of phlebotomy practices, staff training on chain of custody protocols, and inspection of storage and transportation facilities. Quality Control helps ensure that all steps in the phlebotomy process are followed correctly and that the integrity of the samples is preserved throughout the testing process.
Conclusion
Maintaining the chain of custody during phlebotomy procedures is essential to ensure the accuracy and reliability of diagnostic testing. By following specific steps such as proper patient identification, accurate labeling of blood samples, secure storage and transportation, chain of custody documentation, and Quality Control measures, Healthcare Providers in the United States can uphold the integrity of the testing process and deliver precise results to patients.
Disclaimer: The content provided on this blog is for informational purposes only, reflecting the personal opinions and insights of the author(s) on the topics. The information provided should not be used for diagnosing or treating a health problem or disease, and those seeking personal medical advice should consult with a licensed physician. Always seek the advice of your doctor or other qualified health provider regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website. If you think you may have a medical emergency, call 911 or go to the nearest emergency room immediately. No physician-patient relationship is created by this web site or its use. No contributors to this web site make any representations, express or implied, with respect to the information provided herein or to its use. While we strive to share accurate and up-to-date information, we cannot guarantee the completeness, reliability, or accuracy of the content. The blog may also include links to external websites and resources for the convenience of our readers. Please note that linking to other sites does not imply endorsement of their content, practices, or services by us. Readers should use their discretion and judgment while exploring any external links and resources mentioned on this blog.
