Steps and Procedures for Obtaining Informed Consent for Phlebotomy in Medical Labs
Summary
- Obtaining Informed Consent is a critical part of the phlebotomy procedure in medical labs in the United States.
- The steps and procedures for obtaining Informed Consent involve explaining the procedure, risks, benefits, and alternatives to the patient.
- Proper documentation of Informed Consent is essential to ensure that patients understand and agree to the phlebotomy procedure.
Introduction
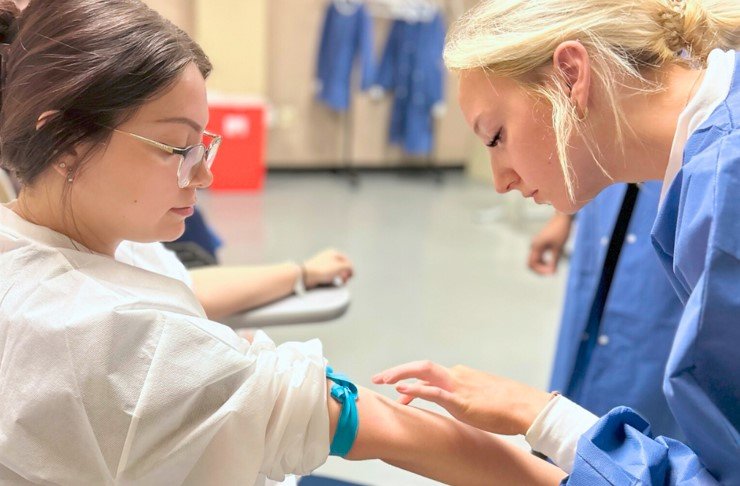
Obtaining Informed Consent is a fundamental ethical and legal requirement in medical practice, including phlebotomy procedures in clinical labs. In the United States, Healthcare Providers must ensure that patients understand the nature of the procedure, potential risks and benefits, and alternatives before obtaining their consent to proceed. In this article, we will discuss the required steps and procedures for obtaining Informed Consent from patients prior to performing a phlebotomy procedure in a medical lab.
What is Informed Consent?
Informed Consent is a process by which Healthcare Providers communicate relevant information to patients so that they can make an informed decision about their care. This process involves explaining the nature of the proposed procedure, including the risks, benefits, and alternatives, in a language that the patient can understand. Informed Consent is based on the principle of autonomy, which respects the right of individuals to make decisions about their own healthcare.
Steps and Procedures for Obtaining Informed Consent for Phlebotomy
Step 1: Explanation of Phlebotomy Procedure
- Before obtaining consent for a phlebotomy procedure, the healthcare provider must explain the nature of the procedure to the patient.
- The explanation should include details such as the purpose of the blood draw, the location of the vein to be accessed, and the equipment that will be used.
- Patients should also be informed about any potential discomfort or pain associated with the procedure.
Step 2: Disclosure of Risks and Benefits
- Healthcare Providers must also disclose the risks and benefits of the phlebotomy procedure to the patient.
- Common risks of phlebotomy include bruising, bleeding, infection, and fainting.
- Benefits of phlebotomy may include diagnosing medical conditions, monitoring treatment effectiveness, and promoting overall health.
Step 3: Discussion of Alternatives
- Patients should be informed about any reasonable alternatives to the phlebotomy procedure.
- Depending on the patient's condition and healthcare goals, alternatives may include non-invasive Diagnostic Tests, imaging studies, or monitoring without blood draws.
- Patients should have the opportunity to ask questions and express their preferences regarding the procedure and alternatives.
Step 4: Patient Understanding and Agreement
- After explaining the procedure, risks, benefits, and alternatives, Healthcare Providers must ensure that the patient understands this information.
- Patients should have the opportunity to ask questions and seek clarification about any aspect of the phlebotomy procedure.
- Once the patient understands the information provided, they can voluntarily agree to the procedure by signing a consent form.
Step 5: Documentation of Informed Consent
- Proper documentation of Informed Consent is essential to ensure that patients have been informed about the phlebotomy procedure and have agreed to it voluntarily.
- Healthcare Providers should document the discussion of the procedure, risks, benefits, alternatives, patient understanding, and agreement in the patient's medical record.
- Consent forms should include details such as the date of the consent, patient's name, healthcare provider's name, and a description of the procedure.
Conclusion
Obtaining Informed Consent is a critical component of the phlebotomy procedure in medical labs in the United States. Healthcare Providers must follow specific steps and procedures to ensure that patients are fully informed about the nature of the procedure, potential risks and benefits, and alternatives before obtaining their consent. Proper documentation of Informed Consent is essential to protect both patients and Healthcare Providers. By following these steps, Healthcare Providers can uphold ethical standards and promote patient autonomy in the phlebotomy process.
Disclaimer: The content provided on this blog is for informational purposes only, reflecting the personal opinions and insights of the author(s) on the topics. The information provided should not be used for diagnosing or treating a health problem or disease, and those seeking personal medical advice should consult with a licensed physician. Always seek the advice of your doctor or other qualified health provider regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website. If you think you may have a medical emergency, call 911 or go to the nearest emergency room immediately. No physician-patient relationship is created by this web site or its use. No contributors to this web site make any representations, express or implied, with respect to the information provided herein or to its use. While we strive to share accurate and up-to-date information, we cannot guarantee the completeness, reliability, or accuracy of the content. The blog may also include links to external websites and resources for the convenience of our readers. Please note that linking to other sites does not imply endorsement of their content, practices, or services by us. Readers should use their discretion and judgment while exploring any external links and resources mentioned on this blog.
