What Forms Are Used for Informed Consent in Phlebotomy and Diagnostic Labs
When patients visit a phlebotomy or diagnostic lab for blood work or medical testing, it is essential that they are fully informed about the procedures being performed and provide their consent before any samples are taken. Informed Consent is a crucial aspect of healthcare Ethics, ensuring that patients understand the risks, benefits, and alternatives to a proposed treatment or procedure. In this article, we will explore the different forms that are used for obtaining Informed Consent in phlebotomy and Diagnostic Labs.
What is Informed Consent?
Informed Consent is a process through which Healthcare Providers communicate information to patients about a proposed medical procedure or treatment. This information includes the nature of the procedure, its risks and benefits, any alternative options, and the patient's right to refuse or withdraw consent at any time. Informed Consent is an essential component of patient autonomy and respect for individual decision-making.
Why is Informed Consent Important in Phlebotomy and Diagnostic Labs?
Obtaining Informed Consent is particularly important in phlebotomy and Diagnostic Labs, where patients may be receiving a variety of blood tests, imaging studies, or other medical procedures. Informed Consent ensures that patients understand what is being done to their bodies, why it is necessary, and what potential risks or complications may be involved. It also gives patients the opportunity to ask questions, seek clarification, and make an informed decision about their healthcare.
Forms Used for Informed Consent in Phlebotomy and Diagnostic Labs
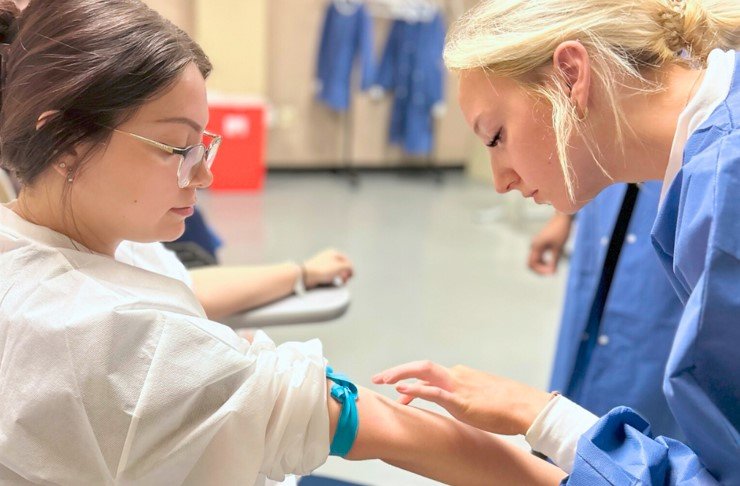
1. Consent Form for Blood Collection
One of the most common forms used in phlebotomy is the consent form for blood collection. This form typically outlines the purpose of the blood test, the specific tests being performed, and any potential risks or discomfort associated with the procedure. It also includes a section where the patient can provide their signature to indicate their voluntary consent to the blood draw.
2. MRI Consent Form
In Diagnostic Labs that perform imaging studies such as MRIs, patients are often required to sign a consent form before the procedure. The MRI consent form typically includes information about the purpose of the test, the use of magnetic fields and radio waves, and any potential risks or contraindications. Patients may also be asked to provide information about medical conditions, allergies, or implants that could affect the safety of the MRI procedure.
3. Biopsy Consent Form
For procedures such as biopsies, a separate consent form is typically required. The biopsy consent form outlines the nature of the procedure, the reason for performing the biopsy, and any potential risks or complications, such as bleeding or infection. Patients are given the opportunity to ask questions and seek clarification before signing the consent form.
4. Laboratory Test Consent Form
When patients are undergoing a series of laboratory tests, they may be asked to sign a general consent form that covers all the tests being performed. This form typically includes information about the purpose of the tests, the types of samples being collected, and any risks or side effects associated with the testing process. By signing the consent form, patients indicate their agreement to the entire battery of tests being performed.
Best Practices for Obtaining Informed Consent
Provide information in a clear and understandable manner.
Use simple language and avoid medical jargon.
Encourage patients to ask questions and seek clarification.
Ensure patients have adequate time to review the information and make a decision.
Respect the patient's right to refuse or withdraw consent at any time.
Conclusion
Informed Consent is a fundamental aspect of patient-centered care, ensuring that individuals are actively involved in their healthcare decisions. In phlebotomy and Diagnostic Labs, obtaining Informed Consent is crucial to promoting patient autonomy, transparency, and trust. By using appropriate consent forms and following best practices for communication and decision-making, Healthcare Providers can uphold the principles of Informed Consent and ensure the well-being of their patients.
Related Videos
Disclaimer: The content provided on this blog is for informational purposes only, reflecting the personal opinions and insights of the author(s) on phlebotomy practices and healthcare. The information provided should not be used for diagnosing or treating a health problem or disease, and those seeking personal medical advice should consult with a licensed physician. Always seek the advice of your doctor or other qualified health provider regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website. If you think you may have a medical emergency, call 911 or go to the nearest emergency room immediately. No physician-patient relationship is created by this web site or its use. No contributors to this web site make any representations, express or implied, with respect to the information provided herein or to its use. While we strive to share accurate and up-to-date information, we cannot guarantee the completeness, reliability, or accuracy of the content. The blog may also include links to external websites and resources for the convenience of our readers. Please note that linking to other sites does not imply endorsement of their content, practices, or services by us. Readers should use their discretion and judgment while exploring any external links and resources mentioned on this blog.
